
Four Drivers of Success in Conducting DFM to Generate Cost-Savings
What is Design for Manufacturing?
Design for Manufacturing (DFM) is the process of designing parts, components, or products for ease of manufacturing with an end goal of making a better product in a cost-efficient way. This is done by simplifying, optimizing, and refining the product design. The acronym DFMA (Design for Manufacturing and Assembly) is sometimes used interchangeably with DFM. The importance is underlined by the fact that approximately 70% of the manufacturing costs of a product (cost of materials, processing, and assembly) are determined by design decisions.
Main Factors Affecting Manufacturability and Driving Costs
(1) Standard Components vs. Tailored Solutions
Leveraging standard components certainly has many advantages and is one of the main cost drivers in the design and development process. Standard components have higher availability and proven reliability which leads to accelerated product consistency that significantly reduces the product lead time. In addition, using standard components makes it easier to manage inventory and mitigates the risk of meeting tight production timelines through reducing the dependency on specific vendors (commodities are available from multiple sources). While standard components come with many advantages, BIT is always looking for the best solution for its clients which requires balancing tailored technologies with ready to use modules.
Example
Over the years, BIT identified key players in the industry and established long-standing partnerships to benefit from their expertise in key areas. One of the most used off-the-shelf components by BIT are stepper motors. Instead of developing a custom solution, BIT leverages the many years of development and manufacturing work of its partner, and receives additional advanced features such as current limiting algorithms and resonance reducing capabilities. By using proven off-the-shelf technologies in key areas, BIT’s clients benefit from additional industry expertise and save on development costs.
(2) Leveraging Injection Molding
The most common use cases leveraging injection molding in IVD instrumentation are for intricate internal parts and external housing. Compared to, for example, blow molding, the major benefit to injection molding is that it allows the incorporation of more features at very small tolerances with unlimited repeatability. Whilst the initial investment is higher compared to other techniques, cost advantages will be generated through a lower piece cost and an increase in the level of production output (effect of economies of scale).
Example
While conducting a DFM analysis of a Point-of-Care (POC) diagnostic platform for a start-up, BIT proposed to leverage injection molding for the instrument housing and internal parts. At the prototype stage, the plastic housing of the client’s platform was manufactured externally using an expensive (over $1,000/ set) polyurethane casting process with a 30-day lead time. By leveraging injection molding BIT was able to significantly reduce the overall unit cost, shorten the lead time for production, and reduce the part cost by 70%.
(3) Die Casting vs. Machining
While there are many effective processes for making metal parts, the two most popular practices are die casting and CNC machining. Die casting refers to the method of melting metal material (aluminum, copper, zinc, etc.) and injecting it into a steel die or mold in the specific shape of the part. Machining, on the other hand, refers to programming a CNC robot to cut the metal in a specific shape. While machining might be more beneficial for low volume parts, since there are no initial tooling costs involved, die casting is the more cost-effective solution for high-volume of consistent and reliable parts.
Example
BIT successfully conducted a DFM analysis for a rapid diagnostic immunoassay device. One of the outcomes to decrease the part costs was to redesign key internal and intricate parts using the die casting process instead of CNC machining. To avoid additional costs, secondary machining was minimized through redesigning interfacing parts while enhancing the overall functionality. The higher initial tooling cost of die casting proved to be the more cost-effective solution through an increase in the level of production output over time. By leveraging die casting, BIT was able to shorten the assembly time per instrument and reduce the part cost by 80%.
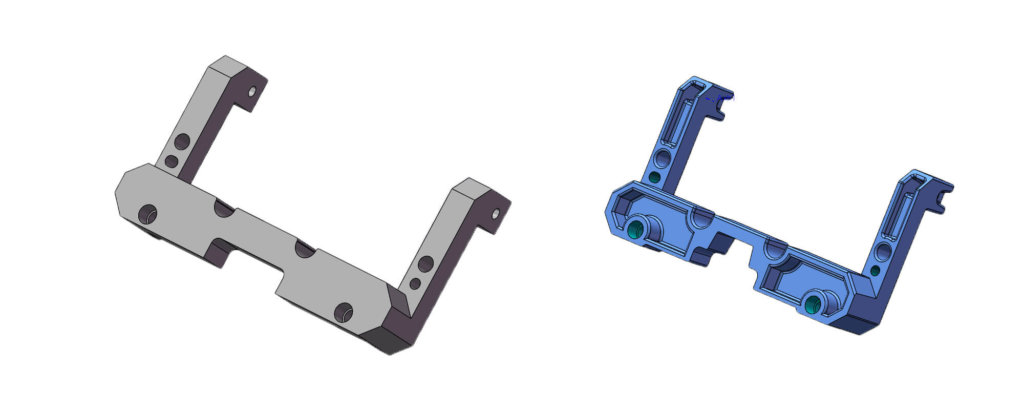
(4) Reducing Number of Parts
Reducing the number of parts, and combining parts wherever possible, is seen as the best opportunity to generate cost savings. Fewer parts imply fewer purchases that translate to less supply chain time, better inventory management, easier material handling, faster processing time, shorter development time, enhanced assembly, faster quality control and inspection, and much more. Overall, the level of intensity of all product-related activities during the device lifecycle gets reduced. Most importantly, this approach results in a better-quality product.
Example
An American IVD client approached BIT with an original instrument design that consisted of four machined parts that were assembled using fasteners. After performing a DFM analysis, BIT created a simplified design with fewer single components (eliminating fasteners) to be more cost-effective. BIT was able to reduce the part cost by 75% and shorten the assembly time per device by over 10min. Additionally, other cost savings were achieved on inventory, material handling, and processing time by using the die casting process for part manufacturing.

BIT’s Manufacturing Background is Key to Development Success
Throughout the 43-year tenure in the medical device and IVD industries, BIT has gained an acute understanding that a “manufacturing mindset” is imperative to a project throughout the entire lifecycle. This includes having manufacturing leaders to be part of the development project team from the beginning. The objective of conducting the DFM is to optimize manufacturability while preserving the functionality of the product. Even though implementing DFM practices on in-vitro diagnostic devices is challenging due to the inherent low volume production, BIT applies the DFM process wherever possible to ensure the product is as cost-competitive as possible to allow our partners to gain early market share. The whole process is supported by BIT’s in-house regulatory team to ensure that all relevant regulatory requirements are achieved.

